
Figure 4 A-F
A, B, Radioactive in vitro kinase assays. Recombinant wildtype and phosphosite mutants of mouse (A) and human (B) FLNc Ig-like domains 23-24 (d23-24) were treated with PKCα in the presence of [γ-33P]ATP and analysed by SDS-PAGE followed by autoradiography or Coomassie staining. S2625 of mFLNC d23-24 was replaced by A or D; S2623/S2624 of hFLNC d23-24 by AA or DD. As a control, PKCα was incubated in [γ-33P]ATP-containing kinase buffer without hFLNc d23-24 (B). WT, wildtype; A, alanine; D, aspartate; +, 10 ng PKCα; ++, 20 ng PKCα
C, D, MS-based in vitro kinase assays. Reactions were performed as described in (A, B) using unlabelled ATP and PKCα. MS data from three independent kinase experiments for mFLNc d23-24 WT, AA and DD sites mutants (C) and mFLNc WT, A, and D site mutants (D) were quantified. Intensities of phosphopeptides distinctive for a specific phosphorylation site (red) were added up per experiment and represented as normalized mean ± SEM.
E, F, Fragmentation spectra of mono-phosphorylated peptides of mouse (E) and human (F) FLNc d23-24 WT forms. PKCα-dependent phosphorylation of mFLNc-S2625 and hFLNc-S2623 was determined by higher-collisional dissociation and electron transfer dissociation, respectively. Fragment ions exhibiting a neutral loss of phosphoric acid (H3PO4; 97.9768 u) are marked with an asterisk (*); loss of water (H2O) as indicated. Phosphorylated residues are depicted in red; b/c- and y/z-ion series in red and blue, respectively.
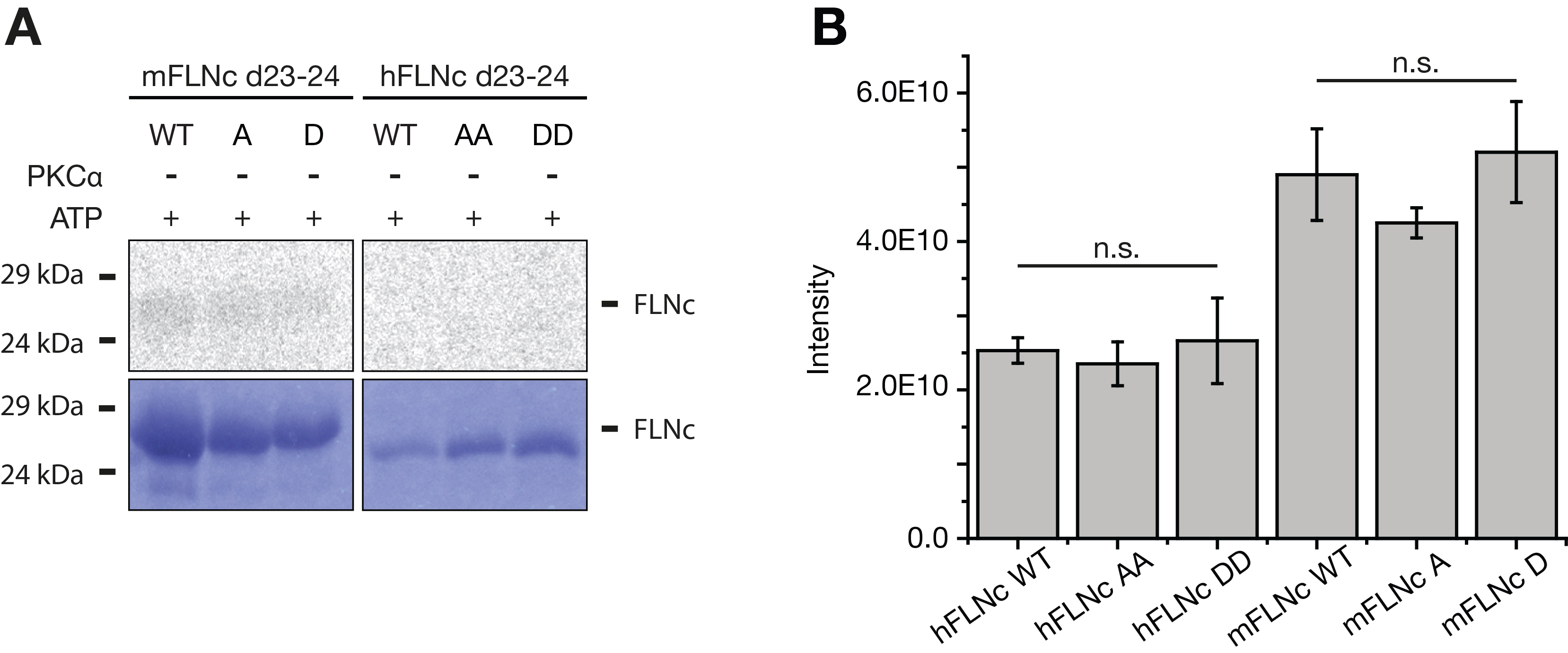
Figure S3.
A, Radioactive in vitro kinase assays. Recombinant wildtype and phosphosite mutants of mouse and human FLNc Ig-like domains 23-24 (d23-24) were treated with [γ-33P]ATP in the absence of PKCα and analysed by SDS-PAGE followed by autoradiography or Coomassie staining. S2625 of mFLNC d23-24 was replaced by A or D; S2623/S2624 of hFLNC d23-24 by AA or DD. As a control, PKCα was incubated in [γ-33P]ATP-containing kinase buffer without hFLNc d23-24 (B). WT, wildtype; A, alanine; D, aspartate
B, Abundance of recombinant mouse and human FLNc d23-24 wildtype and phosphosite mutants in mass spectrometry-based in vitro kinase assays. Shown are the mean values of the total peptide intensity measured for each of the FLNc d23-24 constructs (see Figure S2B). Data derived from three independent experiments per construct. Error bars represent the SEM. WT, wildtype; A, alanine, D, aspartate; n.s., not significant